
To promote talent and potential, the Prices for Master Classes are very affordable. Master Teachers cater to teaching Maths, Physics, Chemistry and Biology (Science) for 6th to 12th grades across CBSE and ICSE Boards. To ensure that motivation is stirred in the best proportion for your clear understanding, a good number of quizzes and Objective tests like V-Brainer, V-Maths, Turbo Maths are organized to impart knowledge and reward the best performers with surprise gifts. Hours and Hours of Study with no fun, is a bad idea for you, foreseeing the long run. Vedantu is the first choice of students aspiring to score full marks in their ICSE and CBSE Board exams or to crack any competitive exam like IIT JEE (Mains & Advanced), Kishore Vaigyanik Protsahan Yojana (KVPY), National Talent Search Exam (NTSE), International Math Olympiad (IMO), International English Olympiad (IEO). Interactive approach establishes a well-deserved academic connect between you and Master Teachers. Sessions get recorded for you to access for quick revision later, just by a quick login to your account. Your academic progress report is shared during the Parents Teachers Meeting. Assignments, Regular Homeworks, Subjective & Objective Tests promote your regular practice of the topics. Revision notes and formula sheets are shared with you, for grasping the toughest concepts. WAVE platform encourages your Online engagement with the Master Teachers. We provide you year-long structured coaching classes for CBSE and ICSE Board & JEE and NEET entrance exam preparation at affordable tuition fees, with an exclusive session for clearing doubts, ensuring that neither you nor the topics remain unattended. We have grown leaps and bounds to be the best Online Tuition Website in India with immensely talented Vedantu Master Teachers, from the most reputed institutions. Vedantu LIVE Online Master Classes is an incredibly personalized tutoring platform for you, while you are staying at your home. So Vm = molar volume of gas = 22.71 L mol-1Įxample: What is the volume occupied by 3.70 moles of N2 gas at STP? How many moles of helium gas are present in the sample?Ĭonditions: STP (standard temperature and pressure, 0☌ and 100 kPa) R – Gas Constant (value depends upon the units of Pressure, Volume and Temperature)Įxample: A sample of pure helium gas occupies a volume of 6.8 L at 0☌ and 100 kPa. N – Number of moles of gas, P – Pressure, T- Temperature In order to calculate molar volume of a substance, we can divide the molar mass by its density. The most common example to illustrate is the molar volume of a gas at STP (Standard Temperature and Pressure), which is equal to 22.4 L for 1 mole of any ideal gas at a temperature equal to 273.15 K and a pressure equal to 1.00 atm. It’s generally applied only to gasses, where the identity of the gas doesn’t affect the volume. In simple words, Molar volume is the Volume occupied by one mole of any substance at a given temperature and pressure. How to Calculate Molar Volume of a Substance & Solved Examples Thus, the Molar Volume Formula| How to Calculate Molar Volume of a Substance| Solved Examples PDF will be very useful for students in their studies. Students can download this article on Molar Volume Formula| How to Calculate Molar Volume of a Substance| Solved Examples on their device in PDF form. 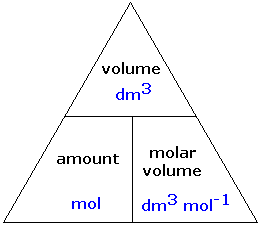
It will be beneficial for students studying for their board exams too. The descriptions provided here will be very beneficial for students for their competitive examination preparation such as NEET or JEE. The expert faculty have had a lot of experience in teaching Chemistry to students. Students can find the Molar Volume Formula given here. 
To explain the method of calculating the How to Calculate Molar Volume of a Substance the help of solved examples has been taken. Here, the process of calculating the Molar Volume of a Substance has been explained very easily. The same value is obtained for all gases at the same temperature and pressure.Students can learn how to find the How to Calculate Molar Volume of a Substance from this website for free. The molar volume is the volume occupied by one mole of any gas.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
